 As discussed above the difference between the electronegativity of Chlorine and Phosphorus atom develops the polarity across the P-Cl bond.Īll the 3 P-Cl bonds are polar having a partial negative charge on chlorine atom and the partial positive charge on Phosphorus atom. Phosphorus trichloride molecule is made up of 3 chlorine and 1 phosphorus atom. You can check out the reason for the non-polarity of CS2. The dipole moment of nonpolar molecules is always equaled to zero.įew examples of nonpolar molecules are CS2, O2, etc. And the geometrical shape of nonpolar molecules is always symmetric due to which the charge distribution on the atoms of these molecules is equally dispersed. The electronegativity of atoms in these molecules is equal in most cases. Nonpolar Molecule: These are the molecules that have an equal distribution of charge on its atoms. You can check out the reason for the polarity of CH2O. Few examples of polar molecules are OF2, CH2O, etc. The dipole moment of polar molecules is always equaled to non zero. It is generally seen that the geometrical shape of polar molecules is asymmetric(distorted) due to which the charge disperses unevenly on the molecule. The electronegativity of atoms in these molecules differs from each other and have an unequal share of bonded electrons. Polar Molecules: The molecules that have unequal distribution of charge on its atoms are polar molecules. Let us check what differences do polar and nonpolar molecules make between them The covalent bonds can be polar and nonpolar depending upon various factors discussed in the below topics. The molecules are held different types of bonds including ionic, covalent, metallic, hydrogen bonding. In addition to this, the electronegativity of Phosphorus is 2.19 and that of Chlorine is 3.19.īeing higher electronegative, chlorine atom attracts the bonded electron pair slightly towards its side and gains partial negative charge and phosphorus atom gains partial positive charge. In the PCl3 molecule, the phosphorus atom is considered to have an oxidation state of +3 and chlorine has an −1 oxidation state. If we talk about the chemical composition of Phosphorus trichloride, It consists of 1 Phosphorus atom and 3 Chlorine atoms.Īll three chlorine atoms are connected to Phosphorus atom via a single covalent bond and complete its octet leaving behind a lone pair on the Phosphorus atom.ĭue to lone pair on Phosphorus atom, the repulsion between lone pair and bond pairs, the shape of the molecule becomes tetrahedral with all 3 chlorine atoms at three vertices of pyramid and Phosphorus leading as the central atom. The molecular mass of the PCl3 molecule is 137.33 g/mol. It is volatile in nature and reacts vigorously with water to produce HCl gas. It is also considered a toxic substance for living beings. Phosphorus(III) chloride and Phosphorous chloride.Īt room temperature, it exists in the liquid state and is colorless to yellowish in appearance. Phosphorus trichloride has its systematic IUPAC name as Trichlorophosphane. So, Is PCl3 Polar or Nonpolar? PCl3 is a polar molecule because of its tetrahedral geometrical shape having a lone pair on Phosphorus atom and the difference between the electronegativity of Chlorine(3.16) and Phosphorus(2.19) atoms resulting in unequal sharing of electrons and develop positive and negative poles across the molecule making it a polar molecule.   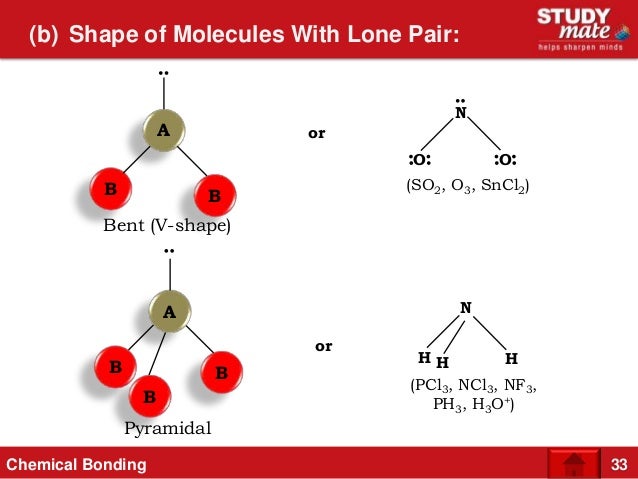 
In this article, I will answer this and will cover its properties and applications. Many of you may have a question regarding whether PCl3 is a polar substance or not. It exists as a colorless to yellow fuming liquid and considered to be toxic in nature. A molecule may be nonpolar either when there is an equal sharing of electrons between the two atoms of a diatomic molecule or because of the symmetrical arrangement of polar bonds in a more complex molecule.Phosphorus trichloride is a chemical substance known by its chemical formula PCl3. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
